
Understand AAV
Our understanding of AAV (ANCA-associated vasculitis) is increasing

Understand AAV
Our understanding of AAV (ANCA-associated vasculitis) is increasing

Understand AAV
Our understanding of AAV (ANCA-associated vasculitis) is increasing
Menu


Introduction to AAV
Incidence & prevalence
AAV is a rare, severe small vessel vasculitis that affects multiple organs and has a high acute mortality risk.1
Types of AAV
The most common subtypes of AAV are granulomatosis with polyangiitis (GPA, previously called Wegener's) and microscopic polyangiitis (MPA).2,3
AAV is a rare disease2
- Global prevalence: 30–218 per million4
- European incidence: 13–20 per million per year2,4
AAV occurrence
- AAV can affect both younger and older people, but is rare in children and young people and incidence rises with age2,5
- AAV occurs slightly more frequently in men (annual incidence rate approximately 54%) than in women2,5

References & footnotes
Abbreviations
AAV, ANCA-associated vasculitis; ANCA, anti-neutrophil cytoplasmic antibody; EGPA, eosinophilic granulomatosis with polyangiitis; GPA, granulomatosis with polyangiitis; MPA, microscopic polyangiitis
References
- Hutton HL, et al. Semin Nephrol 2017;37(5):418–35.
- Watts RA, et al. Nephrol Dial Transplant 2015;30(Suppl 1):i14–22.
- Yates M, et al. Ann Rheum Dis 2016;75(9):1583–94.
- Mohammad AJ. Rheumatology (Oxford) 2020;59:iii42–50.
- Rathmann J, et al. RMD Open 2023;9:e002949
- Watts RA, et al. Rheumatology (Oxford) 2017;56(9):1439–40.
Introduction to AAV
System organ damage
AAV can result in systemic organ damage and failure, with the kidney and lung as major targets1-3
AAV can result in damge to vital organs such as the lungs, kidneys, nervous system, gastrointestinal system, skin, eyes and heart.3,4 Both GPA and MPA show a lot of clinical commonality.2
In a population of 848 patients, the cumulative incidence of end-stage renal disease (ESRD) rose steadily over time, reaching 26.8 at 15 years. Patients who developed ESRD had significantly higher mortality; of the 175 with ESRD, 118 (67.4%) died during follow-up, most of whom were on dialysis (78.6%). Those who progressed to ESRD had a much lower baseline eGFR (13.2 vs. 42.1 mL/min/1.73m²). Although some patients initially requiring renal replacement therapy (RRT) regained function, most relapsed. These findings underscore the importance of early diagnosis and intervention in AAV to prevent irreversible kidney damage.5


A. ENT2
Frequency
GPA: 50–95%; MPA: 2–30%
Manifestations
GPA: Crusting rhinitis, destructive sinusitis, saddle-nose deformity, nasal septum deformity, otitis media MPA: Not specific, not destructive, not granulomatous
B. Lungs2
Frequency
GPA: 60–80%; MPA: 60–80%
Manifestations
GPA: Lung plain and/or excavated nodules, alveolar haemorrhage, bronchial and/or subglottic stenosis
MPA: Alveolar haemorrhage
C. Skin2
Frequency
GPA: 10–50%; MPA: 35–60%
Manifestations
GPA and MPA: Purpura
D. Kidneys2
Frequency
GPA: 60–80%; MPA: 80%
Manifestations
GPA, MPA: Pauci-immune necrotising extra-capillary glomerulonephritis
GPA and MPA patients accumulate organ damage from a combination of vasculitis activity and glucocorticoid-related AEs7-9
Long-term and repeated high-dose glucocorticoid use is associated with an increased risk of new onset/worsening of diabetes mellitus, hypertension, osteoporosis, avascular necrosis of bone, malignancy, cataracts and other debilitating side effects.7,8‡§
High levels of long-term vasculitis damage were independently associated with increased cumulative glucocorticoid use (p=0.016).9§
This serious morbidity is accompanied by a significantly increased long-term mortality risk, with a hazard ratio of 2.41 (95% CI: 1.74-3.34) in GPA patients compared with age- and sex-matched controls.8-10§*
At a mean of 7 years post diagnosis in patients with GPA or MPA…8§

References & footnotes
Footnotes
*AAV patients with (n=212; mean age: 58 years; PR3-ANCA positive: n=138; MPO-ANCA positive: n=65; ANCA negative: n=9) and without (n=61; mean age: 52 years) renal involvement (active urinary sediment, proteinuria, impaired renal function or biopsy) diagnosed and treated with AAV at a single centre in the Netherlands between January 1990 and December 2007 who were followed until death, loss to follow-up or December 2010.5
†Retrospective data collected from patients (n=72; mean age: 64 years) with a clinical AAV diagnosis (Chapel Hill disease definitions and PR3-/MPO-ANCA positive) and renal involvement at diagnosis (rise in serum creatinine >30% and/or erythrocyturia [≥10 red blood cells per high-power field and/or red blood cell casts]) in a single-centre cohort (Northwest Clinics, Netherlands) between 1 February 2005 and 1 February 2015.6
‡Patients (n=524) with newly diagnosed GPA or MPA (adapted Chapel Hill disease definitions) recruited into four EUVAS RCTs in 42 hospitals in 11 European countries and Mexico between 1995 and 2005, and followed for 1 year.7
§Patients (n=735 recruited; mean age: 57.6 years) with newly diagnosed GPA (55%) or MPA (45%; adapted 1994 Chapel Hill disease definitions, clinical presentation, and positive ANCA serology and/or histology) recruited into six EUVAS RCTs between 1995 and 2009. Long-term follow-up data (n=535 eligible patients; GPA: 53%; MPA: 48%; mean age: 57.7 years) were available from four RCTs (n=467/535 with data available; mean follow-up: 7.3 years) with VDI data from 302/535 patients (GPA: 55%; MPA: 45%; mean age: 56.8 years; mean follow-up: 7.1 years) and VDI + glucocorticoid data from 296/535 patients (GPA: 56%; MPA: 44%: mean age: 56.6 years).8,9
**Data collected from patients with a first diagnosis of incident GPA (n=465; mean age: 60.3 years) and non-GPA-matched (sex, age, year of birth and GPA diagnosis year) controls (n=4613; mean age: 60.3 years) from the United Kingdom (The Health Improvement Network) between 1992 and 2013. Mortality data were obtained from the 2003–13 cohort.10
Abbreviations
AAV, ANCA-associated vasculitis; AE, adverse event; ANCA, anti-neutrophil cytoplasmic antibody; CI, confidence interval; ENT, ear, nose and throat; ESRD, end-stage renal disease; EUVAS, European Vasculitis Study Group; GFR, glomerular filtration rate; GI, gastrointestinal; GPA, granulomatosis with polyangiitis; MPA, microscopic polyangiitis; MPO, myeloperoxidase; PR3, proteinase 3; RRT, renal replacement therapy; RCT, randomised controlled trial; VDI, Vasculitis Damage Index
References
- Hutton HL, et al. Semin Nephrol 2017;37(5):418–35.
- Pagnoux C. Eur J Rheumatol 2016;3(3):122–33.
- Kitching A, et al. Nat Rev Dis Primers 2020;6(1):71.
- Al-Hussain T, et al. Adv Anat Pathol 2017;24(4):226–34.
- Sachez-Alamo B, et al. Nephrology Dialysis Transplantation 2024;39(9):1483-93.
- Houben E, et al. BMC Nephrol 2017;18(1):378.
- Little MA, et al. Ann Rheum Dis 2010;69(6):1036−43.
- Robson J, et al. Ann Rheum Dis 2015;74(1):177–84.
- Robson J, et al. Rheumatology (Oxford) 2015;54(3):471–81.
- Wallace ZS, et al. Semin Arthritis Rheum 2016;45(4):483–9.
Introduction to AAV
Mortality & morbidity
AAV leads to an increased risk of mortality, especially in the first year after diagnosis1
In the first year after a GPA, MPA or EGPA diagnosis (n=195), the mortality rate is 13%1*
Of these patients: 27 (28%) of the 98 deaths occurred during the first year after diagnosis with the main causes of death being active vasculitis and bacterial infection1
In a population-based cohort, the cumulative survival rate at 2, 5 and 10 years in patients patients with GPA, MPA or EGPA was 82%, 70% and 55% respectively.1
In the AAV cohort, there was a significant increased mortality compared with the age-matched, sex-matched and calendar year-matched general population.1
In a retrospective cohort study of AAV patients between 2018 and 2025, pulmonary involvement was associated with higher mortality (28.6% vs. 0%) and relapse rates (17.9% vs. 4.5%) compared to those without pulmonary manifestations.2
References & footnotes
Footnotes
*Patients (n=524) with newly diagnosed GPA or MPA (adapted Chapel Hill disease definitions) recruited into four EUVAS RCTs in 42 hospitals in 11 European countries and Mexico between 1995 and 2005, and followed for 1 year.1
†Patients (n=535; median age: 61 years) with newly diagnosed GPA (53%) or MPA (47%; adapted 1994 Chapel Hill disease definitions, clinical presentation, and positive ANCA serology and/or histology) recruited into four EUVAS RCTs in 70 general and university hospitals in 15 countries between 1995 and 2002 with a median 5.16 years of follow-up for all patients. A control cohort was matched for age, sex, year and country.3
‡Data collected from patients with a first diagnosis of incident GPA (n=465; mean age: 60.3 years) and non-GPA matched (sex, age, year of birth and GPA diagnosis year) controls (n=4613; mean age: 60.3 years) from the United Kingdom (The Health Improvement Network) between 1992 and 2013, and divided into two cohorts based on the year of diagnosis (1992–2002 and 2003–2013).2
Abbreviations
AAV, ANCA-associated vasculitis; ANCA, anti-neutrophil cytoplasmic antibody; EGPA, eosinophilic granlomatosis with polyangiitis; EUVAS, European Vasculitis Study Group; GPA, granulomatosis with polyangiitis; MPA, microscopic polyangiitis; RCT, randomised controlled trial
References
- Heijl C, et al. RMD Open. 2017;3:e000435.
- Gökten D, et al. Biomedicines. 2025;13:1401.
Introduction to AAV
Referral, diagnosis & follow-up
AAV patients often experience a complex pathway of patient referral and diagnosis1*
AAV requires multidisciplinary management by centres with, or with ready access to, specific vasculitis expertise.2
The relative rarity and non-specific presentation of AAV can lead to a delay in disease diagnosis of more than 6 months in one-third of patients.3
Diagnosis of AAV and differentiation into the GPA or MPA subtype depends on the patient’s clinical symptom constellation, the results of imaging studies and laboratory investigation.3,4
An organ biopsy, usually renal, is often performed to confirm the diagnosis.3,4




References & footnotes
Footnotes
*Retrospective study reviewing 929 incident AAV patients (GPA: 54%; MPA: 46%; mean age: 57 years; male: 53.7%) in France, Germany, Italy and the United Kingdom (399 physicians) who initiated remission induction therapy between November 2014 and February 2017 with data collected at baseline presentation, and after 1, 3, 6 and 12 months of treatment.1
Abbreviations
AAV, ANCA-associated vasculitis; ANCA, anti-neutrophil cytoplasmic antibody; BMI, body mass index; COPD, chronic obstructive pulmonary disease; ENT, ear, nose and throat; EULAR, European Alliance of Associations for Rheumatology; GP, general practitioner; GPA, granulomatosis with polyangiitis; HIV, human immunodeficiency virus; ICU, intensive care unit; MPA, microscopic polyangiitis; MPO, myeloperoxidase; PR3, proteinase 3
References
- Rutherford PA, et al. J Am Soc Nephrol 2018;29:839(Abstract SA-PO403).
- EULAR Guidelines PLACEHOLDER
- Yates M, Watts R. Clin Med (Lond) 2017;17(1):60–4.
- Pagnoux C. Eur J Rheumatol 2016;3(3):122–33.
Disease mechanism
AAV pathogenesis
The interaction between the activated alternative complement pathway, neutrophils and C5a is associated with vasculitis damage in AAV.1
The development of AAV is a complex and multifactorial autoimmune process2,3
The initial causes of AAV are currently unclear.3 Predisposing factors such as microbial infection, genetic influence, environmental agents and specific drugs are all fundamental to the development of AAV.2,3
Exposure to silica, pesticides, fumes, construction materials, hydrocarbon (cleaning agents, paint, diesel), drugs (propylthiouracil, hydralazine, D-penicillamine, cefotaxime, minocycline, anti-TNF agents, phenytoin) and certain psychoactive agents may all cause AAV.2,3
ANCA involvement
Loss of immune tolerance to ANCA antigens and development of ANCA by plasma cells2
ANCA are most commonly directed against the neutrophil lysosomal enzymes PR3 and MPO in GPA and MPA, respectively2-6
Neutrophils are primed
Neutrophils are primed by inflammatory cytokines (TNF-α, IL-1 and IL-18) produced in response to an infection or another event, with genetic predisposition also relevant3,7,8
ANCA antigens presented
Primed neutrophils present ANCA antigens (e.g. MPO and PR3) at their cell surface that bind to ANCA, resulting in neutrophil activation2,3,7,8
Inflammation mediators released
Activated neutrophils adhere to and penetrate the blood vessel wall, and release mediators of inflammation and cell injury, e.g. NETS2,3,7
Alternative complement pathway activated
Activated neutrophils also release factors such as properdin that have an autocrine role in activating the alternative complement pathway, leading to the generation of C5a2,7
Binding of C5a to C5aR1
Binding of C5a to C5aR1 amplifies ANCA-induced inflammation and vascular damage7
Necrotising vasculitis
This process leads to necrotising vasculitis in small blood vessels7
Chronic inflammation
Over a few days, acute inflammation and necrosis are replaced by chronic inflammation and scarring7
References & footnotes
Abbreviations
AAV, ANCA-associated vasculitis; ANCA, anti-neutrophil cytoplasmic antibody; C5a, complement 5a; GPA, granulomatosis with polyangiitis; IL, interleukin; MPA, microscopic polyangiitis; MPO, myeloperoxidase; NETS, neutrophil extracellular traps; PR3, proteinase 3; R, receptor; TNF, tumour necrosis factor
References
- Bekker P, et al. PLoS One 2016;11(10):e0164646.
- Hutton HL, et al. Semin Nephrol 2017;37(5):418–35.
- Al-Hussain T, et al. Adv Anat Pathol 2017;24(4):226–34.
- Chen M, et al. Medicine (Baltimore) 2008;87(4):203–9.
- Lionaki S, et al. Arthritis Rheum 2012;64(10):3452–62.
- Pagnoux C. Eur J Rheumatol 2016;3(3):122–33.
- Jennette JC, Nachman PH. Clin J Am Soc Nephrol 2017;12(10):1680–91.
- von Borstel A, et al. Autoimmun Rev 2018;17(4):413–21.
Disease mechanism
The importance of complement and neutrophils in AAV activation
C5a plays a major role in the pathogenesis of AAV, amplifying ANCA-induced inflammation and vascular damage1
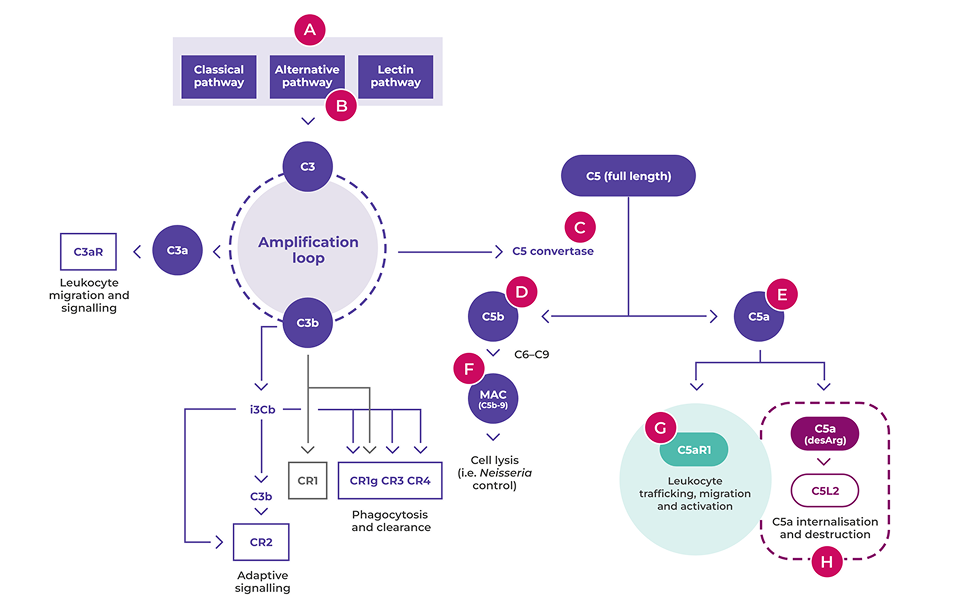

Diagram adapted from Bekker P, et al. PLoS One 2016
A. The three pathways of complement activation (classical, lectin and alternative) result in the formation of C3a, C3b, C5a and C5b-91
B. The alternative complement pathway is constantly ticking over through the spontaneous breakdown of C3 to C3b.2 It is the dysregulation of this pathway that is relevant in AAV1
C. C5 convertase formed during the amplification loop cleaves C5 into C5a and C5b1
D. C5b combines with complement components C6, C7, C8 and C9 to form C5b-9, also known as the terminal complement complex or membrane attack complex1
E. C5a is the downstream terminal effector molecule in the complement cascade and a potent pro-inflammatory mediator critical in AAV1,4
F. C5b-9 is needed for host resistance to encapsulated bacterial infections, such as Neisseria meningitides1,3
G. Binding of C5a to C5aR1 results in two key events that act in a vicious cycle to amplify ANCA-induced inflammation and vascular damage:1,4,5
- Rapid induction of the expression of adhesion molecules on the cell surface that causes leukocyte chemotaxis1
- Further exposure of ANCA antigens at the neutrophil cell surface facilitates subsequent neutrophil activation5
C5aR1 is also thought to play a role in promoting autoimmunity to ANCA antigens andincreasing ANCA production.6
In addition, C5a mediates inflammation by stimulating vascular permeability, neutrophil degranulation, and release of lysosomal proteases and oxidative freeradicals1,7
H. C5L2 activation does not play a role in AAV; in other conditions, it has a currently unclear pattern of pro- and anti-inflammatory actions1,8
C5a is rapidly degraded by removal of the C-terminal arginine, forming C5a des Arg with approximately 10-fold reduced activity on C5aR11,9
C5a des Arg binds with high affinity to the C5L2 receptor, and is subsequently internalised and degraded1,10
References & footnotes
Abbreviations
AAV, ANCA-associated vasculitis; ANCA, anti-neutrophil cytoplasmic antibody; C-terminal, carboxy terminal; MAC, membrane attack complex; R, receptor
References
- Bekker P, et al. PLoS One 2016;11(10):e0164646.
- Thurman JM, Holers VM. J Immunol 2006;176(3):1305–10.
- Figueroa JE, Densen P. Clin Microbiol Rev 1991;4(3):359–95.
- Jennette JC, Nachman PH. Clin J Am Soc Nephrol 2017;12(10):1680–91.
- Jennette JC, et al. Semin Nephrol 2013;33(6):557–64.
- Dick J, et al. Kidney Int 2018;93(3):615–25.
- Gerard C, et al. Annu Rev Immunol 1994;12:775–808.
- Li R, et al. FASEB J 2013;27(3):855–64.
- Monk PN, et al. Br J Pharmacol 2007;152(4):429–48.
- Cain SA, Monk PN. J Biol Chem 2002;277(9):7165–9.
AAV treatment
The balance of treatment
Treatment must balance controlling vasculitis with minimising treatment-related damage1–4
At the time of AAV diagnosis or relapse, the major concerns around treatment involve controlling vasculitis activity and minimising the acute adverse effects from therapy.1,2 Later when remission is achieved, cumulative organ damage, often related to long-term low dose glucocorticoids, and patient experience are more important.1–4
The EULAR Guidelines now state that patients should be periodically screened for treatment-related adverse effects and co-morbidities. They recommend prophylaxis and life-style advice to reduce treatment-related complications and other co-morbidities.5
The toxic affects of treatments can have a profound impact on AAV patients6
Infections, particularly respiratory infections, are a common and important class of complication in patients with AAV and are associated with early mortality.7
Adverse events include short-term complications, such as hyperglycemia and infection (a major cause of early mortality in people with AAV), and long-term complications, including hypogammaglobulinemia and infertility.6
A study showed that infection in the early stage of AAV disease progression increased the risk of death by 2.55 times.7

References & footnotes
Footnotes
*Patients (n=524) with newly diagnosed GPA or MPA (adapted Chapel Hill disease definitions) recruited into four EUVAS RCTs in 42 hospitals in 11 European countries and Mexico between 1995 and 2005, and followed for 1 year.6
†Systematic literature review of 33 studies published between 1 January 2007 and 30 January 2018 containing data on glucocorticoid-related AEs (any untoward medical occurrence) and serious AEs (defined in European Medicines Agency CPMMP/ICH/377/95) which threaten life or function.7
Abbreviations
AAV, ANCA-associated vasculitis; AE, adverse event; ANCA, anti-neutrophil cytoplasmic antibody; EULAR, European Alliance of Associations for Rheumatology; EUVAS, European Vasculitis Study Group; GPA, granulomatosis with polyangiitis; MPA, microscopic polyangiitis; RCT, randomised controlled trial.
References
- Yates M, et al. Ann Rheum Dis 2016;75(9):1583–94.
- Robson JC, et al. Rheumatol Int 2018;38(4):675–82.
- Yates M, Watts R. Clin Med (Lond) 2017;17(1):60–4.
- Robson J, et al. Rheumatology 2015;54(3):471–81.
- Hellmich B, et al. Ann Rheum Dis 2023;0:1–18.
- Titeca-Beauport D, et al. BMC Nephrol. 2018;19 (1):317
- Xu T, et al. RMD Open 2022;8:e002424
AAV treatment

2020
Reducing the GC dose in severe AAV patients did not significantly impact the primary outcome of death or ESRD, but did reduce the rate of infection (HR=0.69)1
- Risk of death or ESRD with reduced GC dose was non-inferior to standard GC dose
- Reduced-dose regimen decreased the risk of serious infections (0.69; 95% CI, 0.52 to 0.93) without increasing the risk of other adverse events
- The continued use of a standard-dose GC regimen, even in patients with severe AAV should be re-evaluated
- Use of PLEX in this patient population provided no added benefit

2020
Extended maintenance therapy leads to better clinical outcomes2
- Relapse rates were reduced in patients receiving long-term RTX
treatment
compared to standard maintenance therapy - With standard maintenance therapy, relapse rates remain high. At month 28, 25% of patients experienced a relapse
- There was no difference in the incidence of AEs with long-term treatment

2019
Preliminary results show that a reduced GC dose can be effective for reinducing remission in AAV, and RTX was superior to AZA at maintaining remission3,4
- Remission induction with a reduced GC dose should be considered clinically, as it was as effective as a typical GC dose regimen
- Initial findings show RTX to be superior to AZA in reducing relapses
- Relapses still occurred in both treatment groups. By 24 months, 13% of the RTX patients and 38% of the AZA patients experienced a relapse

2018
Tailored and fixed-schedule RTX regimens are equally as effective at maintaining remission, but relapses still occur5
- Relapse rates were similar with tailored and fixed-schedule RTX treatment
- The tailored regimen demonstrated that remission maintenance can be achieved with fewer RTX infusions
- Further research is required to establish reliable laboratory tests that can predict the appearance of relapse

2014
RTX is more effective than AZA for maintenance of remission, but GC use and relapse remain common6-9
- RTX was shown to be a more effective maintenance therapy than AZA for newly diagnosed AAV patients
- Minor and major relapses occurred throughout the maintenance phase of treatment. By Month 28, 17% of patients suffered a major relapse, increasing to 38% by Month 60
- GC use remained common amongst all patients throughout the 60-month follow-up period

2010
RTX was found to be non-inferior to daily CYC for the induction and maintenance of remission, but remission rates remain variable10–12
- Patient achievement and maintenance of full remission remains variable with current therapies
- 1 in 3 patients fail to achieve remission at 6 months without use of GCs, and 1 in 2 fail to sustain remission at 12 months
- Non-severe relapse is an under-recognised clinical problem; these patients suffer subsequent relapses resulting in high GC exposure
References & footnotes
Abbreviations
AAV, ANCA-associated vasculitis; AE, adverse event; ANCA, anti-neutrophil cytoplasmic antibody; AZA, azathioprine; CYC, cyclophosphamide; ESRD, end-stage renal disease; GC, glucocorticoid; HR, hazard ratio; PLEX, plasma exchange; RTX, rituximab
References
- Walsh M, et al. N Engl J Med 2020;382(7):622-31.
- Charles P, et al. Ann Intern Med 2020;173(30):179-87.
- Smith RM, et al. Ann Rheum Dis 2020;79(9):1234-96.
- Smith RM, et al. J Am Soc Nephrol 2019;30.
- Charles P, et al. Ann Rheum Dis 2020;173(30)1143-9.
- Guillevin L, et al. N Engl J Med 2014;371(19):1771-80.
- Terrier B, et al. Ann Rheum Dis 2018;77(8):1150-6.
- Terrier B, et al. Ann Rheum Dis 2018;77(8):1150-6. [Supplementary appendix]
- Guillevin L, et al. N Engl J Med 2014;371(19):1771-80. [Supplementary appendix]
- Stone JH, et al. N Engl J Med 2010;363(3):221-32.
- Specks U, et al. N Engl J Med 2013;369(5):417-27.
- Miloslavsky EM, et al. Arthritis Rheum 2015;67(6):1629-36.
Additional resources
NO-AVA-2500029 | Date of preparation: March 2026





